Pharma Is Hollywood, Roughly
How two industries with nothing in common converged on the same business model
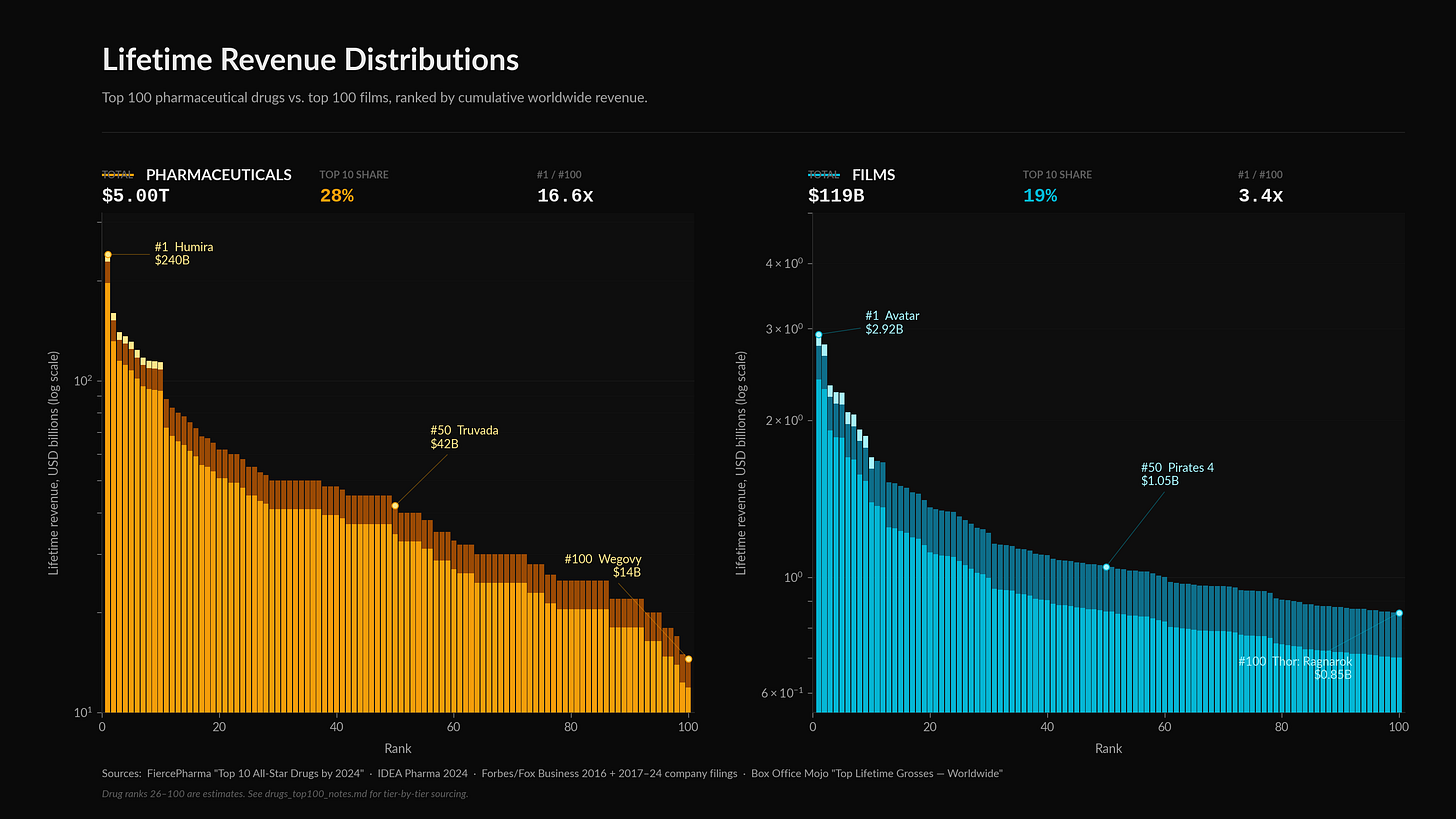
Of all the new drugs introduced in the United States between 1988 and 1992, the top decile by revenue captured 56% of cumulative sales over the next eleven years. Of the 361 new therapeutic agents the FDA approved between 1995 and 2014, the 25 best-sellers - about 7% of the cohort - accounted for 38% of cumulative global revenue. Only about 1 in 8 drugs that reached the market ever generate more than $1 billion in cumulative sales - and most of those that don’t never end up able to recoup development costs.
Hollywood box office is right-skewed in a comparable way. A small number of blockbusters dominate the empirical mean by enough that the median wide-release film barely covers its production cost while the top of the distribution generates billions in worldwide receipts. The mathematical fit has been studied repeatedly since De Vany and Walls (1999) showed that box-office returns follow a heavy-tailed distribution in which a few hits drive most of the revenue. Industry concentration runs in parallel: in 2023, the top three studios by domestic gross (Universal, Warner Bros., Disney) captured well over half of U.S. box office between them, and the top ten releases were dominated by franchise installments.
But pharmaceuticals and movies are absolutely nothing alike. They have completely different products, different customers, and wildly different regulators (the FDA is famously strict about clinical endpoints, whereas nobody has ever been required to run a double-blind placebo-controlled trial to prove that Fast & Furious 10 cures boredom). The buyers, the sellers, and the actual physical things changing hands do not line up in any conceivable way. The unit economics differ by several orders of magnitude, and the development cycles differ by years.
So why on earth do they share remarkably similar revenue distributions?
Not A Coincidence
The first instinct of someone who has read a little economics is to say power laws are everywhere. Wealth is power-law-distributed! City sizes are power-law-distributed! The frequency of the word “the” is power-law-distributed! Big deal, so what.
This is correct but unhelpful. Generic power-law-ish revenue curves happen all the time, and Scherer and Harhoff (2000) showed that innovation returns broadly are heavy-tailed, not just pharma and film. But the specific shape shared by these two industries is much weirder: heavy-tailed payouts generated by a sequential gating process, featuring characteristic abandonment options at every step, all bathed in a thick, unyielding fog of Knightian uncertainty about whether anyone will actually like the thing. That second shape is what both pharma and film have, and it isn’t shared with toothpaste, or industrial chemicals, or smartphones.
The reason Hollywood and Big Pharma end up in the exact same statistical attractor is that they both have the exact same strange economic machinery under the hood:
First, bespoke, one-off production. Every single artifact is entirely unique. There is no The Godfather Part II (well, okay, bad example, but you know what I mean), and there is no second Keytruda. Economists call this the “infinite variety” property of creative goods. The same is true of new chemical entities, where every literal structure is its own patentable snowflake
Second, comically massive sunk costs. A summer blockbuster costs maybe $400 million all-in. An FDA-approved drug runs somewhere between $1 billion (if you are very optimistic) and $2.87 billion (if you ask Tufts). You can only play this game if your organization is completely comfortable piling a billion dollars into a trench, lighting it on fire, and walking away whistling.
Third, sequential gating with binary failure points. A drug has to survive preclinical, then Phase 1, Phase 2, Phase 3, and FDA review. A film has to survive the option, development hell, greenlight, production, and opening weekend. Each gate culls a huge fraction of the population. Good pharma attrition data puts the Phase-1-to-approval rate at around 13.8% (and oncology at a dismal 3.4%). Hollywood attrition rates from optioned script to wide release look roughly identical.
Fourth, Knightian uncertainty about commercial outcomes. I mean the vintage 1921 Frank Knight stuff, where you don’t even know the probability distribution. You cannot just A/B test a novel leukemia drug to see if it catches on, because the A/B test is called a “Phase 3 clinical trial” and costs half a billion dollars. And even after launch, things don't get better: a Nature Reviews Drug Discovery analysis of 1,700 sell-side forecasts found that consensus analyst predictions remained off by a median of 45% even six years after a drug hit the market. Six years of real sales data, and the forecasters still couldn't tell you within half what next year's revenue would be.
Meanwhile, preview screenings for movies are famously terrible predictors of opening weekend (which is itself a terrible predictor of total revenue).
Finally, belief-mediated choice. The mystery of the fruits of both Pharma and Film lives at the exact moment of choosing between comparable things you haven’t tried yet. Keytruda or Opdivo for your melanoma? Zoloft or Prozac for your depression? Star Wars or Star Trek for your Friday night? You can’t test them all, so you blindly outsource the epistemic burden to specialized intermediaries: oncologists, psychiatrists, Rotten Tomatoes, or that one annoying friend who insists you simply must watch Lars von Trier.
That last point does a lot of heavy lifting. Individually, movies and pills are experience goods: you swallow the pill or watch the film, and you see if you feel better. But the counterfactuals are credence goods. Did this SSRI cure my anxiety, or did I just finally delete Twitter? Was this Marvel movie actually the optimal use of my Friday night, or would I have been happier staring at a wall? You can never really know...
When evaluating the counterfactual is impossible, we fall back on reputation. In any industry with a heavy-tailed talent distribution, reputation inevitably follows the Rosen superstar dynamic, where a microscopic bump in perceived quality magically transmutes into a monstrous avalanche of revenue. Let me try to map out exactly how this works.
Movie Critics, But For Drugs
Everyone knows how the Hollywood prestige machine works. You have your Rotten Tomatoes aggregators, Metacritic scores, and festival juries at Cannes and Sundance handing down verdicts of cinematic virtue or whatever. Of course nobody will tell you that the Cannes jury is personally buying millions of movie tickets; instead, they are signaling to the next layer of the ecosystem — i.e. the distributors, exhibitors, and type of people who want to look smart at dinner parties — that a film is officially Good.
Pharma has the Key Opinion Leader or KOL. A KOL is a doctor equipped with heavy academic credentials, a long publication record, and the ability to influence how their peers prescribe drugs. Pharma companies happily pay them to advise on clinical trials, speak at sponsored medical events, author papers, and give the big plenary talks at conferences. Similar to the verdict of a jury at Sundance, the KOL’s blessing convinces the next layer of the bureaucratic onion: e.g. formulary committees, regular prescribing doctors, and the people who write medical guidelines - that a drug is worth using. The actual patient is much too far downstream to be a useful marketing target.
There is one wrinkle. A KOL’s name often appears as the author of the very work being judged - say, on a clinical paper that the pharmaceutical company conveniently helped design, write, and place. This bundles two functions that Hollywood generally tries to keep strictly separate: the gatekeeper-of-quality role (the critic) and the credited-creator role (the director). But a KOL gets to play both. Sergio Sismondo’s Ghost-Managed Medicine (2018) argues that around 40% of recently approved-drug clinical literature is ghost-managed by industry agents - meaning that these studies are designed, written, and placed by pharma employees, with academic KOLs only attached as nominal authors. You can contest the exact percentage here… but the existence of the system is pretty much undeniable. Internal pharma documents released through litigation in the Vioxx, Paxil, and Neurontin cases describe the Publication Planning Industrial Complex in detail.
Sismondo tries to treat this bundling as pharma’s own unique and special pathology, which seems a little unfair. Sell-side equity research has the same problem, too: the analyst rating a stock works at a bank that wants the issuer's investment banking business. Moody's and S&P also get paid by the bond issuers they rate. And the Big Four audit firms are hired and fired by the same companies whose books they sign. I’d even go as far as to say that film critics are not nearly as independent as my Hollywood analogy above suggested; junkets, exclusive access, and embargoed screenings produce their own subtle kinds of capture. Having gatekeepers whose income depends on the firms they evaluate is just what reputation infrastructure looks like in the wild.
If anything, Sismondo’s argument lands much stronger… if you invert it. Pharma isn’t the dirtiest case because its bundling is uniquely egregious. But it looks like the dirtiest case because it is uniquely legible. Under the Sunshine Act, the CMS Open Payments program disclosed $12.75 billion in payments and ownership interests from pharma and device manufacturers to U.S. healthcare professionals in 2023 alone. That was distributed across roughly 15.6 million transactions involving about 940,000 physicians (for context, the entire 2023 U.S. domestic box office was about $9 billion). Pharma spends more money paying doctors to influence other doctors than Hollywood collects from American moviegoers in a whole year! And we only know this because pharma is legally required to report it.
Disanalogies
I do suspect there are some places the structural analogies I’ve been drawing between Pharma and Hollywood break down, which is why we should probably classify pharma and film as “evolutionary cousins” rather than “identical twins”.
First, while the FDA acts as a regulatory gatekeeper, the actual bottleneck for drug quality control largely downstream of biology itself. Hollywood, on the other hand, has a softer principle around this. Because movies are credence goods, critical consensus and commercial success can diverge wildly; "good" is entirely in the eye of the beholder.Venom (2018) got a catastrophic 30% on Rotten Tomatoes and still made $856 million worldwide because it had brand recognition and a great October slot. And it got two sequels after that! Sonic the Hedgehog (2020) survived a horrifying viral CGI-teeth scandal just by redrawing Sonic, ultimately grossing $319 million. The Twilight movies were critically savaged across every conceivable dimension and still became a defining franchise of the 2010s.
Tumors, notoriously, do not grade on a curve. If your shiny new molecule fails to beat placebo in a properly powered double-blind Phase 3 trial, no amount of clever positioning can save you. For most diseases that lack noisy endpoints, biology is a hard physical constraint (the drug either shrinks the mass… or it doesn't!). This is the underlying reason the FDA as a regulatory gate exists at all - they’re enforcing constraints that biology itself imposes in ensuring that they only approve drugs which improve how patients feel/function/survive.
Then there is the matter of intellectual property, which in pharma means “a ticking time bomb” and in Hollywood means “roughly the heat death of the universe.”
Drug patents expire mechanically. You get twenty years from the filing date, plus maybe a few compensatory extensions if the FDA took exceptionally long to read your paperwork. The moment that clock runs out, generic competitors flood the zone and your revenue evaporates overnight. AbbVie’s Humira went from making $14.4 billion in 2023 to $9.0 billion in 2024. That is a 37.6% drop in a single year, caused entirely by the sudden materialization of U.S. biosimilars.
The upshot is that drug franchises decay on a strictly enforced, rather predictable schedule. This essentially means that all forward facing pharma companies are stuck on a grueling treadmill of finding the Next Big Thing, forced to constantly invent or acquire novel chemical entities just to replace the ones plummeting off the patent cliff.
Film copyrights, meanwhile, last for the life of the author plus seventy years, or ninety-five years if you are a corporation. Steamboat Willie only just tumbled into the public domain in 2024, roughly a century after he first whistled. Barring a sudden collapse of the U.S. legal system, Disney will still be extracting rents from their current crop of Marvel and Star Wars characters long after everyone reading this is dead.
The third disanalogy is the distribution channel. Movies are sold directly to people with eyeballs i.e. the signal flows in something vaguely resembling a free market.
Pharmaceuticals, on the other hand, are sold through a baroque, Lovecraftian B2B nightmare. The patient does not pay full price; the insurer pays most of it; the insurer’s pharmacy benefit manager negotiates rebates with the manufacturer, often via a confidential side-payment that bears no transparent relationship to list price; the formulary committee decides which drugs are covered at which tier; the prior-authorization office decides which patients can access which drugs; the prescribing physician makes the actual prescription decision under conditions of imperfect knowledge about the patient’s coverage. Deep breath.
This is why pharma marketing budgets are entirely directed at intermediaries. Studio marketing targets the consumer; pharma marketing targets the people who tell the consumer what to swallow.
So the revenue distributions match and the superstar economics match. But the regulators, the IP regimes, and the distribution channels are otherwise alien to one another. It is a very good analogy, don’t get me wrong, but it only gets you about 80% of the way there, and then politely asks you to get out of the car.
Sequel Sickness
And yet — even with all these disanalogies — the two industries appear to be threatened by the same species of productivity decline.
If you chart the pharmaceutical industry’s ability to invent new drugs per billion inflation-adjusted dollars, the line goes down so predictably that in 2012, Jack Scannell and colleagues formally named the phenomenon. They called it “Eroom’s Law” - Moore’s Law spelled backwards, since R&D productivity has halved every nine years since 1950.
If you look closely, his diagnoses for the causes of Eroom’s Law maps weirdly well onto the movie business:
Better than the Beatles. Every new tentpole competes with thirty years of Marvel, two trilogies of Star Wars, three of Lord of the Rings, and an essentially infinite back catalog on streaming for $9 a month.
Cautious gatekeeper. Hollywood doesn't have an FDA, but studio greenlight processes have ratcheted toward conservatism over the same period. Before a script gets made today, it has to survive an escalating gauntlet of internal expected-value models, brand-safety vetting, and franchise-compatibility filters.
Throw money at it. Marvel's average production budget roughly doubled in a decade — from the $140M class of Phase 1 to the $250M+ class of Phase 4 — with no obvious improvement in critical or commercial outcomes per dollar.
Brute force. CGI-heavy spectacle and franchise IP have crowded out auteur-driven mid-budget originals. All ten of 2024's top-grossing films worldwide were sequels - the first time in at least fifty years.
A year before Scannell published Eroom’s Law, Pammolli and colleagues argued that pharma's productivity decline reflects “an increasing concentration of R&D investments in areas in which the risk of failure is high” - i.e. the industry has chosen its own decline by chasing harder and harder problems. Hollywood has done exactly this, retreating to the most expensive, franchise-dependent corners of the map. I wrote about the consequences of this incentive structure last year in my post on Novartis.
So if you accept the now-conventional diagnosis of what ails pharma — too many me-too drugs, not enough first-in-class novel mechanisms, and an unwillingness to take real scientific risk on smaller programs — you arrive at the diagnosis Steven Soderbergh has been making for Hollywood since his 2013 State of Cinema address: too many tentpoles, not enough mid-budget originals, not enough willingness to take real artistic risk on smaller films.
Mutating into a Studio
Given all the structural overlap between the two, it was probably only a matter of time before someone tried to explicitly run a Hollywood-esque playbook on biotech.
Enter Roivant Sciences. Founded in 2014 by Vivek Ramaswamy, Roivant pitched itself to investors as a sort of Berkshire Hathaway of biotech. The gimmick was the “Vants” - a swarm of subsidiaries each built around a single drug or therapeutic area, complete with their own management teams and distinct capital structures. Axovant did Alzheimer’s, Myovant did women’s health, Dermavant did dermatology, and so on until you presumably run out of medical prefixes.
The basic strategy was to sift through Big Pharma's discarded assets, buy up abandoned drug candidates, place each one inside a bespoke corporate shell, and take them public. If the drug works, yay! You make billions. If it fails, at least the blast radius is safely contained within a single expendable shell company. You could read this as exactly how multi-picture production houses work: financing each project on its own merits so investors can bet on one movie without subsidizing the rest of the slate.
Roivant's first major test of this model was Axovant. In late 2014, they bought intepirdine — a 5-HT6 receptor antagonist that GlaxoSmithKline had given up on after four separate trials — for $5 million. Six months later, Axovant went public, raised $315 million in what was then the largest biotech IPO in history, and briefly boasted a $3 billion market cap despite having exactly eight employees.
Naturally, in 2017, intepirdine failed its Phase 3 trial — to nobody's particular surprise, since GSK had already thrown it in the trash and rival drugs from Lundbeck and Pfizer using the exact same mechanism were failing simultaneously. To stretch the Hollywood analogy here a bit: Axovant essentially bought the script for a movie every other studio had already passed on, threw a massive budget at it, and predictably ended up producing the biological equivalent of the cinematic disaster Cats. The stock vaporized 75% of its value in a day, the company pivoted to gene therapy, and the whole thing was quietly put out of its misery by 2023.
So was Axovant just a fluke, or is the whole Roivant model defective?
Neither, really. The entire point of the Vant structure is that you don’t have to pick winners - merely that you have to assemble a portfolio where one winner is big enough to pay for all the losers. Intepirdine was a bad individual bet (which is exactly why GSK had thrown it out and why three competing 5-HT6 antagonists were failing at the same time), but Roivant didn’t need intepirdine to work - only one Vant, eventually, big enough to cover the next forty failures.
Since intepridine, the externally verifiable wins of the Roivant model are hard to argue with: eight FDA approvals, a $3 billion Sumitomo deal in 2019 for a stake in five of the Vants, and a $7.1 billion sale of Telavant to Roche in October 2023. Roivant’s total market cap across the Vants now sits around $18 billion. The company also reports 12 positive Phase 3 trials since 2019 and more than 40 medicines in development; if you take those numbers at face value, hit rate per asset roughly tracks the industry baseline.
It’s a pattern that generalizes beyond Roivant rather nicely. BridgeBio, Royalty Pharma, and the Great Biotech SPAC bubble are all just variations on this particular theme of risk management; a mirror of Hollywood’s slate-financing structured studios like Legendary or Skydance. And it even somewhat holds in AI, where Google and Amazon sometimes trade equity for compute capacity (essentially asset-level financing for individual training runs).
Crabs All The Way Down
So, there seems to be a very specific economic genus here. Let’s call them “Knightian project industries.” To get into the club, you need four things:
Bespoke, project-level production with high per-project sunk cost.
Knightian uncertainty about commercial outcomes.
Phase-gated development, where you constantly have to decide whether to cut your losses.
Outcome quality hard to assess prospectively, with capital flows through specialized evaluators (critics, KOLS).
If you check all four boxes, congratulations, you are a Knightian project industry. Pharma and Hollywood are both charter members. Other card-carrying members include frontier LLMs, AAA video games, wildcat oil drilling, and venture capital.
Really, what we are gesturing at is a sort of economic carcinization among this slate of businesses within this category. You know how completely unrelated marine animals keep evolving into crabs? Markets do the same thing too. If you mix massive upfront costs with total Knightian uncertainty about whether the biology/audience/oil well geology will actually cooperate: the market will ruthlessly select for a specific corporate anatomy that maximizes chance of survival. You get heavy tails, bundled gatekeepers, asset-level financing, and a few lucky selectors eating all the profits.
Any firm that refuses to undergo this transformation and tries to maintain some fragile, non-crustacean business model simply runs out of runway and starves. The crabs, meanwhile, will be perfectly fine, happily scuttling sideways across the bones of their competitors while clutching a billion-dollar box office sweep in one claw and a blockbuster FDA approval in the other.